Effect of menbutone on oxidative stress in sheep
Abstract
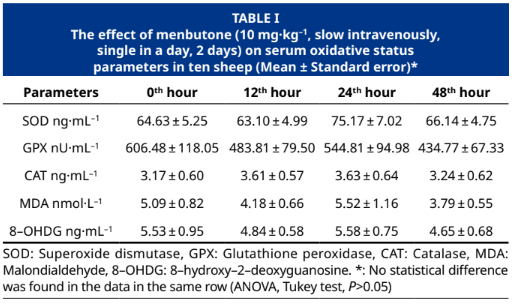
The objective of this research was to determine the effects of administering the choleretic-effective menbutone (genabilic acid) at the recommended dose and duration of serum oxidative status and biochemical parameter levels in sheep. As is known, changes in the oxidative status of living organisms can be observed in many drug applications, disease states, or metabolic disorders. In the study, menbutone was administered at a dose of 10 mg·kg–1 (slow IV, SID, 2 days) to 10 Central Anatolian Merino sheep. Blood samples were collected from the sheep before (0th hour) and at the 12th, 24th, and 48th hours after menbutone administration. Serums were obtained from the collected blood samples. Serum oxidative status parameters (8–hydroxy–2–deoxyguanosine, malondialdehyde, superoxide dismutase, glutathione peroxidase, catalase), troponin I, and creatine kinase-MB isoenzyme levels were measured using the ELISA method, while levels of serum routine biochemical parameters were measured using an autoanalyzer. Menbutone did not affect oxidative stress parameters (8–hydroxy–2–deoxyguanosine, malondialdehyde, superoxide dismutase, glutathione peroxidase, catalase) in sheep (P>0.05) but caused temporary increases (P<0.05) in serum triglyceride and cholesterol levels. However, no statistically significant changes were detected in other serum biochemical parameters (Troponin I, creatine kinase-MB isoenzyme, alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase, low-density lipoprotein, high-density lipoprotein, blood urea nitrogen, creatinine). In conclusion, it can be asserted that menbutone can be used safely in sheep without altering oxidative stress parameters, but monitoring serum triglycerides and cholesterol may be important in animals with metabolic disorders.
Downloads
References
European Medicines Agency (EMA). 16th Meeting of Committee for Veterinary Medicinal Products. [Internet]. The european agency for the evaluation of medicinal products. London (UK): European Medicines Agency. 1996 [cited 24 Mar 2025]. Available in: https://goo.su/pEXDwn
Tras B. Sindirim sistemi ilaçları [Digestive system drugs]. In: Yazar E, editor. Veteriner İlaç Rehberi ve Terapötik El Kitabı [Veterinary Drug Guide and Therapeutic Handbook]. Ankara (Türkiye): Nobel Medical Bookstores; 2024. p. 243–283. Turkish.
Hayvan Bilgi Sistemi-Tarbil [Animal Information System Tarbil]. [Internet]. 2025 [cited 16 Mar 2025]. Turkish. Available in: https://goo.su/4mMV6qQ
Punzo A, Silla A, Fogacci F, Perillo M, Cicero AFG, Caliceti C. Bile acids and bilirubin role in oxidative stress and inflammation in cardiovascular diseases. Diseases [Internet]. 2024; 12(5):103. doi: https://doi.org/qr9c DOI: https://doi.org/10.3390/diseases12050103
Yazar E, Tras B. Serbest oksijen radikalleri, antioksidan enzimler ve antibiyotikler [Free oxygen radicals, antioxidant enzymes and antibiotics]. J. Turk. Vet. Med. Assoc. 2002; 14:42–44. Turkish.
Tabakoglu E, Durgut R. Veteriner hekimlikte oksidatif stres ve bazı önemli hastalıklarda oksidatif stresin etkileri [Oxidative stress in veterinary medicine and effects in some important diseases] AVKAE Derg. [Internet]. 2013 [cited 15 Mar 2025]; 3(1):69–75. Turkish. Available in: https://goo.su/U3bzfHs
Sezer K, Serbest oksijen radikallerinin hastalıkların patogenezisindeki rolü [Role of the free oxygen radicals on the pathogenesis of the diseases]. FU. Sag. Bil. Vet. Derg. [Internet]. 2014 [cited 12 Oct 2025]. 28(1):49–56. Turkish. Available in: https://goo.su/UqSXW9s
Calyniuk B, Grochowska-Niedworok E, Walkiewicz KW, Kawecka S, Popiołek E, Fatyga E. Malondialdehyde (MDA) product of lipid peroxidation as marker of homeostasis disorders and aging. Ann. Acad. Med. Siles. [Internet]. 2016; 70:224–228. doi: https://doi.org/g99sxz DOI: https://doi.org/10.18794/aams/65697
Di Minno A, Turnu L, Porro B, Squellerio I, Cavalca V, Tremoli E, Di Minno MN. 8–hydroxy–2–deoxyguanosine levels and cardiovascular disease: a systematic review and meta- analysis of the literature. Antioxid. Redox Signal. [Internet]. 2016; 24(10):548–555. doi: https://doi.org/f8fx2h DOI: https://doi.org/10.1089/ars.2015.6508
Pilger A, Rüdiger HW. 8–hydroxy–2–deoxyguanosine as a marker of oxidative DNA damage related to occupational and environmental exposures. Int. Arch. Occup. Environ. Health. [Internet]. 2006; 80:1–15. doi: https://doi.org/b6r433 DOI: https://doi.org/10.1007/s00420-006-0106-7
Saracoglu A, Temel HE, Ergun B, Colak O. Oxidative stress- mediated myocardiotoxicity of ciprofloxacin and ofloxacin in juvenile rats. Drug Chem. Toxicol. [Internet]. 2009; 32(3):238–242. doi: https://doi.org/c2jzzx DOI: https://doi.org/10.1080/01480540902882176
Coskun D, Parlak K, Dik B, Faki HE, Bahcivan E, Yazar E, Er A. Effect of enrofloxacin on the joint fluid/blood oxidative status and organ damage markers. Annu. Res. Rev. Biol. [Internet]. 2018; 25(3):1–7. doi: https://doi.org/qr9d DOI: https://doi.org/10.9734/ARRB/2018/40537
Coskun D, Canbar R, Korkmaz Y, Dik B, Er A, Yazar E. Determination of the effect of danofloxacin on 8–hydroxy–2– deoxyguanosine level. Eurasian J. Vet. Sci. [Internet]. 2019; 35(4):224–229. doi: https://doi.org/qr9f DOI: https://doi.org/10.15312/EurasianJVetSci.2019.223
Yazar E. Veteriner İlaç ve Aşı – A dan Z ye (2018 – 2019) [Veterinary Drugs and Vaccines – A to Z (2018 – 2019). Ankara (Türkiye): Nobel Medical Bookstores; 2018. 306 p. Turkish.
Kaya S, Ünsal A. İlaçların istenmeyen etkileri [Adverse effects of medications]. In: Kaya S, Pirinçci İ, Bilgili A, editors. Veteriner Hekimliğinde Farmakoloji [Veterinary Applied Pharmacology]. 3rd ed. Ankara (Türkiye): Medisan; 2002. p.142–152.
Dogan F. Pharmacovigilance in veterinary profession. Eurasian J. Vet. Sci. [Internet]. 2011 [cited 15 Mar 2025]; 27(1):19–25. Available in: https://goo.su/qkWms
Corum O, Er A, Dik B, Eser H, Bahcivan E, Yazar E. Determination of the safety of tulathromycin in sheep. Eurasian J. Vet. Sci. [Internet]. 2015; 31(3):152–157. doi: https://doi.org/qr9g DOI: https://doi.org/10.15312/EurasianJVetSci.2015310972
Corum O, Dik B, Bahcivan E, Eser H, Er A, Yazar E. Cardiac safety of gamithromycin in ewes. Eurasian J. Vet. Sci. [Internet]. 2016; 32(4):242–245. doi: https://doi.org/qr9h
Kerr MG. Veterinary Laboratory Medicine. Clinical biochemistry and haematology. 2nd ed. London (UK): Blackwell Science; 2002. 386 p. Available in: https://goo.su/ozVrL DOI: https://doi.org/10.1002/9780470690246
Turgut K. Karaciğer testi (Liver test). In: Turgut K, editor. Veteriner Klinik Laboratuvar Teşhis [Veterinary Clinical Laboratory Diagnostics]. Konya (Türkiye): Gardeners Printing House; 2000. p. 202–257.
Turgut K. Endokrin, metabolik ve lipid bozukluklarının testleri [Tests for endocrine, metabolic, and lipid disorders]. In: Turgut K, editor. Veteriner Klinik Laboratuvar Teşhis [Veterinary Clinical Laboratory Diagnostics]. Konya (Türkiye): Gardeners Printing House; 2000. p. 416–486.
Er A, Corum O, Eser H, Bahcivan E, Dik B, Yazar E. Effect of dexamethasone treatment on blood oxidative status and prostaglandin F2α metabolite levels in ram. Eurasian J. Vet. Sci. [Internet]. 2016; 32(2):89–93. doi: https://doi.org/qsbs DOI: https://doi.org/10.15312/EurasianJVetSci.2016422395
Ider M, Naseri A, Parlak TM, Zhunushova A, Yazar E. Safety of an antiprotozoal drug combination in sheep. Eurasian J. Vet. Sci. [Internet]. 2020; 36(2):115–120. doi: https://doi.org/qsbt DOI: https://doi.org/10.15312/EurasianJVetSci.2020.268
Canbar R, Uslu M, Arslan MS, Yazar E. Effect of combined application of ivermectin and praziquantel on oxidative stress and selected biochemical parameters in sheep. Eurasian J. Vet. Sci. [Internet]. 2023; 39(1):25–29. doi: https://doi.org/qsbv DOI: https://doi.org/10.15312/EurasianJVetSci.2023.395
Cobrin AR, Blois SL, Kruth SA, Abrams-Ogg ACG, Dewey C. Biomarkers in the assessment of acute and chronic kidney diseases in the dog and cat. J. Small Anim. Pract. [Internet]. 2013; 54(12):647–655. doi: https://doi.org/f5htzf DOI: https://doi.org/10.1111/jsap.12150
El Okle SO, Tohamy GH, Lebda AM. Evaluation of acute toxicity of genabilic acid (menbutone 10%) in rabbits. World Rabbit Sci. [Internet]. 2014; 22(3):215–222. doi: https://doi.org/qsbw DOI: https://doi.org/10.4995/wrs.2014.1791
Slowikowska M, Siwinska N, Zak A, Borowicz H, Kubiak K, Niedzwiedz A. Myonecrosis in a horse after an intramuscular injection of menbutone. Med. Weter. [Internet]. 2018; 74(12):795–798. doi: https://doi.org/qsbx DOI: https://doi.org/10.21521/mw.5991
Lukanc B, Pogorevc E, Kastelic A, Erjavec V. Retrograde jejunal intussusception in a one-year-old cat after treatment with metoclopramide and menbutone. Slov. Vet. Res. [Internet]. 2014 [cited 15 Mar 2025]; 51(4):201–207. Available in: https://goo.su/iFQhUFV DOI: https://doi.org/10.26873/SVR-24675-2014