Histological evaluation of the effects of systemic administration of strontium ranelate on bone healing in rat tibia fractures
Abstract
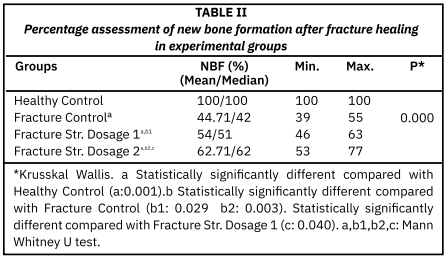
This study aimed to investigate the effects of strontium renelate, a known bisphosphonate-like agent with effects on bone tissue, on bone fracture and defect healing using histological methods. For this purpose, a fracture study was conducted using 28 rats, each consisting of healthy controls (n = 7), fracture controls, and treatment groups, receiving strontium dose 1 (450 mg/kg three times per week), and strontium dose 2 (900 mg/kg three times per week). After a six-week fracture healing period, tibia bones were subjected to histological analysis for new bone formation. Data were analyzed using Kruskal Wallis and Mann Whitney U tests. New bone formation was significantly lower in the fracture groups compared to healthy controls (P < 0.001). An increase in new bone formation ratios was observed in the strontium ranelate-administered groups compared to the fractured controls (P< 0,05). New bone formation was significantly higher in the high-dose strontium ranelate group compared to the low-dose strontium ranelate group (P < 0,05). When histological evaluations and numerical analyses were evaluated together, it was concluded that systemic strontium ranelate administration significantly accelerated new bone formation and maturation processes during fracture healing, depending on the dose used (450 and 900 mg/kg).
Downloads
References
Padilla-Eguiluz NG, Gómez-Barrena E. Epidemiology of long bone non-unions in Spain. [Internet]. Injury. 2021; 52(4):3-7. doi: https://doi.org/mhsc DOI: https://doi.org/10.1016/j.injury.2021.02.053
Santolini Santo E, West R, Giannoudis PV. Risk factors for long bone fracture non-union: a stratification approach based on the level of the existing scientific evidence. Injury. [Internet]. 2015; 46(8):8-19. doi: https://doi.org/f8hdxj DOI: https://doi.org/10.1016/S0020-1383(15)30049-8
Wier J, Shelby H, Bergren S, Patterson JT, Lieberman JR. Modern Approaches and Emerging Biological Therapies to Treat Fracture Nonunion. Pharmaceutics. [Internet]. 2025; 17(11):1457. doi: https://doi.org/qw9c DOI: https://doi.org/10.3390/pharmaceutics17111457
Chun YS, Lee DH, Won TG, Kim Y, Shetty AA, Kim SJ. Current Modalities for Fracture Healing Enhancement. Tissue Eng. Regen. Med. [Internet] 2022; 19(1):11-17. doi: https://doi.org/qw9d DOI: https://doi.org/10.1007/s13770-021-00399-0
Dang Y, Zhang Y, Luo G, Li D, Ma Y, Xiao Y, Xiao L, Wang X. The decisive early phase of biomaterial-induced bone regeneration. Appl. Mater. Today [Internet]. 2024; 38:102236. doi: https://doi.org/qw9f DOI: https://doi.org/10.1016/j.apmt.2024.102236
Staszak K, Rzelewska-Piekut M, Regel-Rosocka M. Role of noble and rare earth metals in bioactive materials for medical applications in tissue engineering. RSC Adv. [Internet]. 2025; 15(48):40709-40729. doi: https://doi.org/qw9g DOI: https://doi.org/10.1039/D5RA04036A
Li K, Liu L, Zhang J, Liao C, Hu J, Song J. TP508 Promotes Bone Regeneration on Distraction Osteogenesis via the Activation of Wnt/ -catenin Signaling Pathway. Curr. Pharm. Biotechnol. [Internet]. 2025; 26(3):402-410. doi: https://doi.org/qw9h DOI: https://doi.org/10.2174/0113892010289575240306033011
Alatabi HSH, Tobji S, Haouas Z. Effects of Calcitonin Administration on the Amount of Bone Formation After Sutural Expansion Using Micro-CT. J. Craniofac. Surg. [Internet] 2023; 34(8):2556-2559. doi: https://doi.org/qw9j DOI: https://doi.org/10.1097/SCS.0000000000009575
Straub J, Sewing A, Walter N, Wong RMY, Alt V, Heiss C, Rupp M. Calcium Sulfate Bone Substitutes in Clinical Use: History, Material Properties, Application, and Outlook for the Future. J. Biomed. Mater. Res. B Appl. Biomater. [Internet] 2025;113(4):e35555. doi: https://doi.org/qw9k DOI: https://doi.org/10.1002/jbm.b.35555
Schortinghuis J, Bronckers AL, Gravendeel J, Stegenga B, Raghoebar GM. The effect of ultrasound on osteogenesis in the vertically distracted edentulous mandible: a double-blind trial. Int. J. Oral Maxillofac. Surg. [Internet]. 2008; 37(11):1014-1021. doi: https://doi.org/cw8qsr DOI: https://doi.org/10.1016/j.ijom.2008.07.004
Kołodziejska B, Stępień N, Kolmas J. The Influence of Strontium on Bone Tissue Metabolism and Its Application in Osteoporosis Treatment. Int. J. Mol. Sci. [Internet]. 2021; 22(12):6564. doi: https://doi.org/qw9m DOI: https://doi.org/10.3390/ijms22126564
Marx D, Rahimnejad-Yazdi A, Papini M, Towler M. A review of the latest insights into the mechanism of action of strontium in bone. Bone Rep. [Internet]. 2020; 12:100273. doi: https://doi.org/grtcqh DOI: https://doi.org/10.1016/j.bonr.2020.100273
Gusman DJ, Matheus HR, Alves BE, Ervolino E, de Araujo NJ, Piovezan BR, Fiorin LG, de Almeida JM. Influence of systemic strontium ranelate on the progression and as adjunctive therapy for the nonsurgical treatment of experimental periodontitis. J. Clin. Exp. Dent. [Internet]. 2021; 13(12):1239-1248. doi: https://doi.org/qw9n DOI: https://doi.org/10.4317/jced.58827
You J, Zhang Y, Zhou Y. Strontium Functionalized in Biomaterials for Bone Tissue Engineering: A Prominent Role in Osteoimmunomodulation. Front. Bioeng. Biotechnol. [Internet]. 2022; 10:928799. doi: https://doi.org/gtptgs DOI: https://doi.org/10.3389/fbioe.2022.928799
Wang S, Xia D, Dou W, Chen A, Xu S. Bioactive Porous Composite Implant Guides Mesenchymal Stem Cell Differentiation and Migration to Accelerate Bone Reconstruction. Int. J. Nanomedicine. [Internet]. 2024; 19:12111-12127. doi: https://doi.org/qw9p DOI: https://doi.org/10.2147/IJN.S479893
Hu Y, Huang J, Chen C, Wang Y, Hao Z, Chen T, Wang J, Li J. Strategies of Macrophages to Maintain Bone Homeostasis and Promote Bone Repair: A Narrative Review. J. Funct. Biomater. [Internet]. 2023; 14(1):18 doi: https://doi.org/g9bg5f DOI: https://doi.org/10.3390/jfb14010018
Falgayrac G, Farlay D, Ponçon C, Béhal H, Gardegaront M, Ammann P, Boivin G, Cortet B. Bone matrix quality in paired iliac bone biopsies from postmenopausal women treated for 12 months with strontium ranelate or alendronate. Bone. [Internet]. 2021; 153:116107. doi: https://doi.org/qw9r DOI: https://doi.org/10.1016/j.bone.2021.116107
Byeon SM, Bae TS, Lee MH, Ahn SG. Guided bone regeneration of calcium phosphate-coated and strontium ranelate-doped titanium mesh in a rat calvarial defect model. J. Periodontal Implant Sci. [Internet]. 2024; 54(5):336-348. doi: https://doi.org/qw9t DOI: https://doi.org/10.5051/jpis.2303000150
Markel DC, Dietz PR, Wu B, Chen L, Bou-Akl T, Shi T, Ren W. Repair of a rat calvaria defect with injectable strontium (Sr)-doped polyphosphate dicalcium phosphate dehydrate (P-DCPD) ceramic bone grafts. J. Biomed. Mater. Res. B Appl. Biomater. [Internet]. 2024; 112(2):35388. doi: https://doi.org/qw9v
Abbas AM, Gobran HG, Soliman AEH. Histological evaluation of bone regeneration after topical application of strontium ranelate gel in critical size bone defects in the tibia of induced diabetic rats. Al-Azhar J. Dent. Sci. [Internet]. 2024; 27(4):511-522. doi: https://doi.org/qw9w DOI: https://doi.org/10.21608/ajdsm.2022.170375.1378
Matvieienko LM, Matvieienko RY, Fastovets 00. Effects Of Strontium Ranelate On Alveolar Bone In Rats With Experimental Diabetes Mellitus. Wiad Lek. [Internet]. 2022; 75(1 pt 2):151-155. PMID: 35182114. Available in: https://goo.su/ByKAn8q DOI: https://doi.org/10.36740/WLek202201201
Itting PT, Kruppke B, Hanke T, Vijayan V, Heiss C, Lips KS. BDNF improves the survival of mesenchymal stem cells cultured on pre-structured gelatin material containing strontium and calcium phosphates for bone regeneration. Front. Bioeng. Biotechnol. [Internet]. 2025; 13:1596846. doi: https://doi.org/qw9x DOI: https://doi.org/10.3389/fbioe.2025.1596846
Tao R, Mi B, Hu Y, Lin S, Xiong Y, Lu X, Panayi AC, Li G, Liu G. Hallmarks of peripheral nerve function in bone regeneration. Bone Res. [Internet]. 2023; 11:6 doi: https://doi.org/hbg8r7 DOI: https://doi.org/10.1038/s41413-022-00240-x
Sandoval-Castellanos AM, Claeyssens F, Haycock JW. Bioactive 3D Scaffolds for the Delivery of NGF and BDNF to Improve Nerve Regeneration. Front. Mater. [Internet]. 2021; 8:734683 doi: https://doi.org/qw92 DOI: https://doi.org/10.3389/fmats.2021.734683
Wu Q, Hu L, Yan R, Shi J, Gu H, Deng Y, Jiang R, Wen J, Jiang X. Strontium-incorporated bioceramic scaffolds for enhanced osteoporosis bone regeneration. Bone Res. [Internet]. 2022; 10(1):55. doi: https://doi.org/qw93 DOI: https://doi.org/10.1038/s41413-022-00224-x
Yi J, Li M, Zhu J, Wang Z, Li X. Recent development and applications of electrodeposition biocoatings on medical titanium for bone repair. J. Mater. Chem. B. [Internet]. 2024; 12(39):9863-9893. doi: https://doi.org/qw94 DOI: https://doi.org/10.1039/D4TB01081G
Ozturan KE, Demir B, Yucel I, Cakıcı H, Yilmaz F, Haberal A. Effect of strontium ranelate on fracture healing in the osteoporotic rats. J. Orthop. Res. [Internet]. 2011; 29(1):138-142. doi: https://doi.org/cqd9c5 DOI: https://doi.org/10.1002/jor.21204
Gonçalves FC, Mascaro BA, Scardueli CR, de Oliveira GJPL, Spolidorio LC, Marcantonio RAC. Strontium ranelate improves post-extraction socket healing in rats submitted to the administration of bisphosphonates. Odontology. [Internet]. 2022; 110(3):467-475. doi: https://doi.org/qw95 DOI: https://doi.org/10.1007/s10266-021-00678-1
Yang J, Guo X, Cui Z, Guo H, Dong JN. Efficacy and safety of denosumab and teriparatide versus oral bisphosphonates to treat postmenopausal osteoporosis: a systematic review and meta-analysis. Front. Endocrinol. [Internet]. 2024; 15:1431676. doi: https://doi.org/qw96 DOI: https://doi.org/10.3389/fendo.2024.1431676
Markel DC, Dietz PR, Wu B, Chen L, Bou-Akl T, Shi T, Ren W. Repair of a rat calvaria defect with injectable strontium (Sr)-doped polyphosphate dicalcium phosphate dehydrate (P-DCPD) ceramic bone grafts. J. Biomed. Mater. Res. B Appl. Biomater, [Internet]. 2024; 112(2):35388. doi: https://doi.org/qw9v DOI: https://doi.org/10.1002/jbm.b.35388
Mehta D, Gentleman E. Advances in Strontium-Releasing Biomaterials for Bone Repair. Biomaterials. [Internet]. 2026; 326:123718. doi: https://doi.org/hbh2m4 DOI: https://doi.org/10.1016/j.biomaterials.2025.123718