Occurrence and protective determinants of ruminal lesions in slaughtered beef cattle: Rumen pH and papilla morphology
Abstract
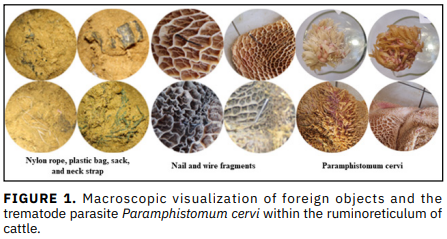
Metabolic imbalances and mechanical trauma induced ruminal lesions are major economic and welfare concerns for the cattle industry. The aims were to characterize rumen fermentation and consider feeding duration, presence of foreign bodies, and fermentation products in relation to lesion formation. The aim of this study was to assess the association between morphological risk factors related to nutrition and reticula-rumen lesions as well as foreign bodies by post-mortem examination in 85 slaughtered cattle, with the resulting data analyzed through multiple logistic regression models. Rumen pH was the only variable with one of the lowest likelihood values for lesions (OR = 0.024; P < 0.001). More specifically, when the rumen pH increased by one unit, the likelihood of lesions decreased by roughly 97.6 %. Protection was also significantly associated with rumen acetic acid concentration (OR= 0.95; P < 0.05). On the other hand, a decrease of 1-unit in acetic acid concentration resulted in an odd ratio approximately 1.053-fold higher for the development of lesion. Also, foreign bodies (14.12 % of cattle were positive) were affected by papilla width in the negative direction coefficient (OR = 0.118; P < 0.05). Decreasing the papilla width by 1 cm increased the chance of identifying a foreign body 8.475-fold more than to overrule one. The presence of lesions and macroscopic ventral sac deformations also showed a strong positive correlation. The results of this study emphasize the necessity for a stable rumen pH and maintenance of epithelial integrity, especially papilla width which may play a major role in reducing ruminal health hazards in cattle.
Downloads
References
Plaizier J C, Krause DO, Gozho GN, McBride BW. Subacute ruminal acidosis in dairy cows: The physiological causes, incidence and consequences. Vet. J. [Internet]. 2008; 176(1):21-31. doi: https://doi.org/c4bdpf DOI: https://doi.org/10.1016/j.tvjl.2007.12.016
Pokhrel B, Jiang H. Postnatal growth and development of the rumen: integrating physiological and molecular insights. Biol. [Internet]. 2024; 13(4):269. doi: https://doi.org/g95qns DOI: https://doi.org/10.3390/biology13040269
Membrive CMB. Anatomy and physiology of rumen. In: Millen DD, Arrigoni MDB; Pacheco RDL (Eds.). Rumenology. (1st Edn.). 2016. Cham, Switzerland: Springer International.1-38. doi: https://doi.org/qvkp DOI: https://doi.org/10.1007/978-3-319-30533-2_1
Penner GB, Steele MA, Aschenbach JR, McBride BW. Ruminant Nutrition Symposium: Molecular adaptation of ruminal epithelia to highly fermentable diets. J. Anim. Sci. [Internet]. 2011; 89(4):1108-1119. doi: https://doi.org/bn2hzp DOI: https://doi.org/10.2527/jas.2010-3378
Hernández J, Benedito JL, Abuelo A, Castillo C. Ruminal acidosis in feedlot: from etiology to prevention. Sci. World J. [Internet]. 2014; 2014(1):702572. doi: https://doi.org/gb5364 DOI: https://doi.org/10.1155/2014/702572
Streitenberger N, Ramiro R, Navarro MA, Asin J, Henderson E, Gonzales-Viera O, Mete A, Torri EH, Uzal FA. Pathology of ruminal acidosis in cattle. Vet. Pathol. [Internet]. 2025; 62(6):939-948. doi: https://doi.org/qvkq DOI: https://doi.org/10.1177/03009858251339889
Umphrey JE, Staples CR. General anatomy of the ruminant digestive system. University of Florida Cooperative Extension Service, Institute of Food and Agriculture Sciences. [Internet]. 1992 [cited 05 Jan 2026]; Available in: https://goo.su/0t5790p
Ramaswamy V, Sharma HR. Plastic bags–Threat to environment and cattle health: A retrospective study from Gondar City of Ethiopia. IIOAB J. [Internet]. 2011 [cited 05 Jan 2026]; 2(1):6-11. Available in: https://goo.su/DSjiLDE
Abdelaal AM, Maghwry SL. Selected studies on foreign body impaction in goats with special reference to ultrasonography. Vet. World. [Internet]. 2014; 7(7):522527. doi: https://doi.org/qgqf DOI: https://doi.org/10.14202/vetworld.2014.522-527
Jonsson NN, Ferguson HJ, Koh-Tan HHC, McCartney CA, Cernat RC, Strachan EM, Thomson W, Snelling TJ, Harvey CD, Andonovic I, Michie C, Wallace RJ. Postmortem observations on rumen wall histology and gene expression and ruminal and caecal content of beef cattle fattened on barley-based rations. Animals. [Internet]. 2020; 14(7):1447-1460. doi: https://doi.org/qvkr DOI: https://doi.org/10.1017/S1751731119002878
Viana PRL, Viana LF, Araújo GHM, Moraes IDTD, Queiroz PJB, Cagnini DQ, Da Silva LAF, Rabelo RE. The macroscopic and microscopic description of ruminal lesions in feedlot bovine. Cienc. Anim. Bras. [Internet]. 2022; 23:e-73109. doi: https://doi.org/qvks DOI: https://doi.org/10.1590/1809-6891v23e-73109p
Luna-Méndez A, Gutiérrez-Chávez A, Valencia-Posadas M, Ruíz-Ramírez J, Pérez-Guiot A, García-Márquez L. Ruminal lesions in cattle slaughtered in slaughterhouses. Abanico Vet. [Internet]. 2020; 10:1-14. doi: https://doi.org/g8p6c3
Wanapat M, Pimpa O. Effect of ruminal NH3-N levels on ruminal fermentation, purine derivatives, digestibility and rice straw intake in swamp buffaloes. Asian-Austral. J. Anim. Sci. [Internet]. 1999; 12(6):904-907. doi: https://doi.org/qvkv DOI: https://doi.org/10.5713/ajas.1999.904
Vargas JAC, de Araújo TC, Mezzomo R. Extraction, identification, and quantification of volatile fatty acids (VFA) in rumen fluid samples using reverse phase highperformance liquid chromatography with diode array detector (RP HPLC-DAD). Res. Sq. [Internet]. 2020; 1–11. doi: https://doi.org/qjgf DOI: https://doi.org/10.21203/rs.3.pex-1121/v1
Markham, P. A steam distilation apparatus suitable for micro-kjeldahl analyses. Biochem. J. [Internet]. 1942; 36(10-12):790-791. doi: https://doi.org/qjgc DOI: https://doi.org/10.1042/bj0360790
Statistical Analysis System SAS. SAS/STAT Software: Hangen and Enhanced, Version 9.4. Cary, North Carolina, USA: SAS Institute.. 2020.
Gönenci R, Yildirim M. Investigation of foreign bodies and their complications in rumen and reticulum of the cattle brought to Iskenderun Slaughterhouse. Vet. Fak. Derg. [Internet]. 2008 [cited 06 Jan 2026]; 19(2):31-36. Available in: https://goo.su/WuRPUy
Yildirim, D. Studying Foreign Bodies at the Rumen and Retikulums of the Cattle Brought to Diyarbakir Ergani Slaughterhouse. [Master's Thesis]. Harran Üniversitesi, Saglik Bilimleri Enstitüsü, Sanliurfa, Türkiye. [Internet]. 2015 [cited 06 Jan 2026]. Available in: https://goo.su/vQitj
Melaku S, Addis M. Prevalence and intensity of Paramphistomum in Ruminants slaughtered at Debre-zeit industrial abattoir, Ethiopia. Global Vet. [Internet]. 2012 [cited 06 Jan 2026]; 8(3):315-319. Available in: https://goo.su/Rusff
Aciöz M, Çeliksöz A, Özçelik S, Degerli S. Prevalence of Paramphistomum spp. in cattle in Sivas province. Medit. Vet. J. [Internet]. 2016; 1(2):7-10. doi: https://doi.org/qvkx DOI: https://doi.org/10.24880/maeuvfd.287345
Singh RP, Sahai BN, Jha GJ. Histopathology of the duodenum and rumen of goats during experimental infections with Paramphistomum cervi. Vet. Parasitol. [Internet]. 1984; 15(1):39-46. doi: https://doi.org/bv9vcm DOI: https://doi.org/10.1016/0304-4017(84)90108-0
Humer E, Petri RM, Aschenbach JR, Bradford BJ, Penner GB, Tafaj M, Südekum KH, Zebeli Q. Invited review: Practical feeding management recommendations to mitigate the risk of subacute ruminal acidosis in dairy cattle. J. Dairy Sci. [Internet]. 2018; 101(2):872-888. doi: https://doi.org/gcwvgm DOI: https://doi.org/10.3168/jds.2017-13191
Dijkstra J, Ellis JL, Kebreab E, Strathe AB, López S, France J, Bannink A. Ruminal pH regulation and nutritional consequences of low pH. Anim. Feed Sci. Technol. [Internet]. 2012; 172(1-2):22-33. doi: https://doi.org/fzftgv DOI: https://doi.org/10.1016/j.anifeedsci.2011.12.005
Luo J, Ranadheera CS, King S, Evans C, Baines S. In vitro investigation of the effect of dairy propioni bacteria on rumen pH, lactic acid and volatile fatty acids. J. Integr. Agric. [Internet]. 2017; 16(7):1566-1575. doi: https://doi.org/gbpp9n DOI: https://doi.org/10.1016/S2095-3119(16)61556-3
Beauchemin KA, Yang WZ, Rode LM. Effects of particle size of alfalfa-based dairy cow diets on chewing activity, ruminal fermentation, and milk production. J. Dairy Sci. [Internet]. 2003; 86(2):630-643. doi: https://doi.org/ffm23s DOI: https://doi.org/10.3168/jds.S0022-0302(03)73641-8
Dragolici V. Rumen parakeratosis intensively fattened young sheep clinical and experimental. Proc. Rom. Acad. Series B. [Internet]. 2014 [cited 12 Oct 2025]; 16(1):55–61. Available in: https://goo.su/ZLzsxf7 DOI: https://doi.org/10.1016/j.anscip.2025.04.042
Diao Q, Zhang R, Fu T. Review of strategies to promote rumen development in calves. Animals [Internet]. 2019; 9(8):490. doi: https://doi.org/p5ks DOI: https://doi.org/10.3390/ani9080490
Dieho K, Dijkstra J, Schonewille JT, Bannink A. Changes in ruminal volatile fatty acid production and absorption rate during the dry period and early lactation as affected by rate of increase of concentrate allowance. J. Dairy Sci. [Internet]. 2016; 99(7):5370-5384. doi: https://doi.org/f8s98j DOI: https://doi.org/10.3168/jds.2015-10819