Primer estudio integral de las poblaciones de garrapatas Ixodidae en caballos Árabe-Berbé del noreste de Argelia: Inventario de especies e influencia de factores abióticos
Resumen
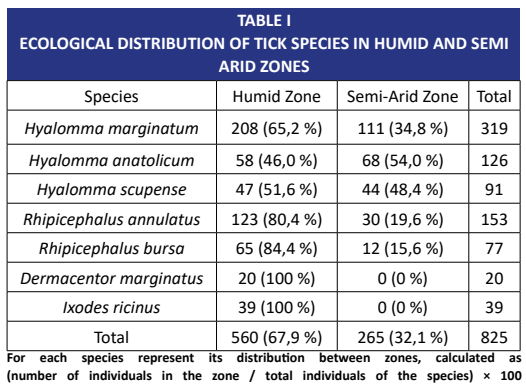
La infestación por garrapatas ha demostrado ser una amenaza real para la salud de los caballos en la cuenca mediterránea. Para identificar los factores ambientales subyacentes a este riesgo, se llevó a cabo un estudio prospectivo de abril a septiembre de 2024 en 67 caballo árabe-bereber distribuidos en seis emplazamientos climáticos y geográficos diferentes, que abarcaban desde las húmedas llanuras costeras de la provincia de El Tarf (4 emplazamientos, n = 46, altitud 50-300 m) hasta las altiplanicies interiores semiáridas de la provincia de Tébessa (2 emplazamientos, n = 21, altitud 400-700 m). Se recolectaron manualmente un total de 825 garrapatas, según un protocolo estandarizado de inspección de cinco minutos, y se diferenciaron microscópicamente mediante un análisis morfológico detallado de características específicLos resultados mostraron que algo más de un tercio de los caballos estaban infestados (frecuencia porcentual global: 34.3 %), con una carga parasitaria media de 36 garrapatas por animal. De manera más significativa, la distribución de especies de garrapatas reveló una clara estructura ecológica (P < 0.001). Las especies del género Rhipicephalus dominaban ampliamente en las áreas húmedas y de baja altitud, representando el 86.2% del total de garrapatas recolectadas. Sin embargo, en las zonas más secas y de mayor altitud, Hyalomma anatolicum fue la especie predominante, representando el 54,0 % de todas las garrapatas recolectadas. Esta diferencia estuvo fuertemente ligada a la altitud, como lo evidenciaron las correlaciones estadísticas: se observó una relación negativa para R. annulatus (ρ = -0.79, P = 0 .002) y R. bursa (ρ = -0.83, P = 0.001), mientras que se encontró una correlación positiva para H. anatolicum (ρ = 0.67, P = 0.015). El modelo estadístico que integraba únicamente la altitud y la zona bioclimática explicó el 69 % de la varianza observada, demostrando así el papel decisivo de estos factores abióticos. Sin embargo, el sexo del caballo (yegua vs. macho) no mostró ninguna relación significativa con el riesgo de infestación (p = 0.741).
Descargas
Citas
Nadal C, Chanet C, Delaunay C, Pitel PH, Marsot M, Bonnet SI. Risk factors for tick infestation and equine piroplasmosis infection among draught horses in France. Ticks Tick Borne Dis. [Internet]. 2025; 16(3):102468. doi: https://doi.org/g9bwcm DOI: https://doi.org/10.1016/j.ttbdis.2025.102468
Kleissl L, Weninger S, Winkler F, Ruivo M, Wijnveld M, Strobl J. Ticks’ tricks: immunomodulatory effects of ixodid tick saliva at the cutaneous tick-host interface. Front. Immunol. [Internet]. 2025; 16:1520665. doi: https://doi.org/g9f72b DOI: https://doi.org/10.3389/fimmu.2025.1520665
Kamran K, Ali A, Villagra C, Siddiqui S, Alouffi AS, Iqbal A. A cross-sectional study of hard ticks (Acari: Ixodidae) on horse farms to assess the risk factors associated with tick- borne diseases. Zoonoses Public Health. [Internet]. 2021; 68(3):247–262. doi: https://doi.org/qtrx DOI: https://doi.org/10.1111/zph.12809
Mota RA, Silito IS, Martins MP, de Oliveira PRF, de Melo RPB, da Silva VV, Jorge FR, Martins TF, Labruna MB. Serological evidence of Rickettsia spp. in horses and survey of tick-borne agents in ticks from horses and wildlife in the state of Pernambuco, Brazil. J. Med. Entomol. [Internet]. 2026; 63(1):tjaf155. doi: https://doi.org/qtrz DOI: https://doi.org/10.1093/jme/tjaf155
Ullah A, Geng M, Chen W, Zhu Q, Shi L, Zhang X, Akhtar MF, Wang C, Khan MZ. Effect of parasitic infections on the hematological profile and reproductive and productive performances in equines. Animals. [Internet]. 2025; 15(22):3294. doi: https://doi.org/qtr3 DOI: https://doi.org/10.3390/ani15223294
Shahzad S, Akinsulie OC, Idris I, Devnath P, Ajagbe D, Aliyu VA, Oladoye MJ, Ukauwa C, Ugwu CE, Ajulo S, Oyeleye BS, Ikele CG, Shelly SY. Ticks and tick-borne diseases in Global South countries: impact and implications of environmental changes. Front. Trop. Dis. [Internet]. 2025; 6:1597236. doi: https://doi.org/qtr4 DOI: https://doi.org/10.3389/fitd.2025.1597236
Cao B, Bai C, Wu K, La T, Chen W, Liu L, Zhou X, Chen C, Li X, Su Y, Che L, Li G. Ticks jump in a warmer world: Global distribution shifts of main pathogenic ticks are associated with future climate change. J. Environ. Manage. [Internet]. 2025; 374:124129. doi: https://doi.org/qtr5 DOI: https://doi.org/10.1016/j.jenvman.2025.124129
Ogden NH, Kotchi SO, Brazeau S, Bouchard C, White JC, Wulder MA, Davidson A, Beaudoin A, Werle D. Tick- borne diseases. In: Brazeau S, Ogden NH, editors. Earth Observation, Public Health and One HealthActivities, Challenges and Opportunities. [Internet]. Wallingford (UK): CABI Publishing; 2022 [cited 15 Oct 2025]. p. 26–37. Available in: https://goo.su/zP5Cp
Boulanger N, Iijima H, Doi K, Watari Y, Kwak ML, Nakao R, Wikel S. Ticks and tick-borne diseases in the northern hemisphere affecting humans. Front. Microbiol. [Internet]. 2025; 16:1632832. doi: https://doi.org/qtsc DOI: https://doi.org/10.3389/fmicb.2025.1632832
Gharbi M, Drissi G, Darghouth MA. Population dynamics of ticks infesting horses in north-west Tunisia. Rev. Sci. Tech. [Internet]. 2018; 37(3):837–841. doi: https://doi.org/qtsd DOI: https://doi.org/10.20506/rst.37.3.2890
Mechouk N, Mihalca AD, Deak G, Bouslama Z. Synopsis of the ticks of Algeria with new hosts and localities records. Parasit. Vectors. [Internet]. 2022; 15(1):302. doi: https://doi.org/qtsf DOI: https://doi.org/10.1186/s13071-022-05424-2
Gholmohammadi S, Malekifard F, Yakhchali M. Study on species distribution and seasonal dynamics of equine tick infestation in Ardabil province, northwest of Iran. Vet. Parasitol. Reg. Stud. Reports. [Internet]. 2024; 48:100987. doi: https://doi.org/qtsg DOI: https://doi.org/10.1016/j.vprsr.2024.100987
Choi H, Lee CH. The impact of climate change on ecology of tick associated with tick-borne diseases. PLoS Comput. Biol. [Internet]. 2025; 21(4):e1012903. doi: https://doi.org/qtsn DOI: https://doi.org/10.1371/journal.pcbi.1012903
Nuttall PA. Climate change impacts on ticks and tick- borne infections. Biologia. [Internet]. 2022; 77:1503–1512. doi: https://doi.org/qtsp DOI: https://doi.org/10.1007/s11756-021-00927-2
Mebarki M, Kaidi R, Benhenia K. Morphometric description of Algerian Arab-Barb horse. Rev. Méd. Vét. [Internet]. 2018 [cited 13 Oct 2025]; 169(7-9):185-190. Available from: https://goo.su/fpxBV
Lilak AA, Matulis G, Potter AM, Pecor DB, Rodriguez D, Wood L, Cerimele R, Stone K, Cleary NG, Butler K, Linton YM, von Fricken ME. A compilation of ticks and tick- borne pathogen distributions in seven countries within North and West Africa from 1901 to 2022: a systematic literature review. Parasit. Vectors. [Internet]. 2025; 19:13. doi: https://doi.org/qtsq DOI: https://doi.org/10.1186/s13071-025-07153-8
Kratou M, Belkahia H, Selmi R, Ben-Abdallah M, Tayh G, Messadi L, Ben-Said M. Molecular identification, genotyping and phylogenetic analysis of Ixodes and Rhipicephalus ticks and their associated spotted fever group Rickettsia species from a single location in northern Tunisia. Front. Microbiol. [Internet]. 2025; 16:1644524. doi: https://doi.org/qtsr DOI: https://doi.org/10.3389/fmicb.2025.1644524
Makwarela TG, Seoraj-Pillai N, Nangammbi TC. Tick Control Strategies: Critical Insights into Chemical, Biological, Physical, and Integrated Approaches for Effective Hard Tick Management. Vet. Sci. [Internet]. 2025; 12(2):114. doi: https://doi.org/qtss DOI: https://doi.org/10.3390/vetsci12020114
Estrada-Peña A, Mihalca AD, Petney TN. Ticks of Europe and North Africa: a guide to species identification. [Internet]. Cham, Switzerland: Springer; 2018 [cited 13 Feb 2026]. doi: https://doi.org/qtst DOI: https://doi.org/10.1007/978-3-319-63760-0
Apanaskevich DA, Horak IG. The genus Hyalomma Koch, 1844. IX. Redescription of all parasitic stages of H. (Euhyalomma) impeltatum Schulze & Schlottke, 1930 and H. (E.) somalicum Tonelli Rondelli, 1935 (Acari: Ixodidae). Syst. Parasitol. [Internet]. 2009; 73(3):199-208. doi: https://doi.org/fkjf9h DOI: https://doi.org/10.1007/s11230-009-9190-x
Bush AO, Lafferty KD, Lotz JM, Shostak AW. Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol. [Internet]. 1997; 83(4):575-583. doi: https://doi.org/dx5kjs DOI: https://doi.org/10.2307/3284227
Benchikh-Elfegoun MC, Kohil K, Gharbi M, Afoutni L, Benachour ML. Cinétique d’infestation par les tiques des bovins de la région subhumide de Constantine en Algérie. Rev. Elev. Med. Vet. Pays Trop. [Internet]. 2019; 72(1):41–45. doi: https://doi.org/qts2 DOI: https://doi.org/10.19182/remvt.31726
Walker AR, Bouattour A, Camicas JL, Estrada-Peña A, Horak IG, Latif AA, Pegram RG, Preston PM. Ticks of domestic animals in the Mediterranean region: a guide to identification of species. Edinburgh, Scotland, UK: Bioscience Reports; 2014 [cited 13 Oct 2025]. Available in: https://goo.su/mnPJ3OX
Benchikh-Elfegoun MC, Gharbi M, Djebir S, Kohil K. Dynamique d’activité saisonnière des tiques ixodidés parasites des bovins dans deux étages bioclimatiques du nord-est algérien. Rev. Elev. Med. Vet. Pays Trop. [Internet]. 2013; 66(4):117–122. doi: https://doi.org/qts4 DOI: https://doi.org/10.19182/remvt.10150
Bouattour A, Darghouth MA, Daoud A. Distribution and ecology of ticks (Acari: Ixodidae) infesting livestock in Tunisia: an overview of eight years field collections. Parassitologia. [Internet]. 1999; 41(Suppl1):510. PMID: 11071534. Available in: https://goo.su/jU5I
Deshpande G, Beetch JE, Heller JG, Naqvi OH, Kuhn KG. Assessing the influence of climate change and environmental factors on the top tick-borne diseases in the United States: a systematic review. Microorganisms. [Internet]. 2024; 12(1):50. doi: https://doi.org/g89nxk DOI: https://doi.org/10.3390/microorganisms12010050
Kim Y, Jaulhac B, Vesga JF, Zilliox L, Boulanger N, Edmunds WJ, Métras R. Environmental drivers of Ixodes ricinus tick population dynamics: mechanistic modelling using longitudinal field surveys and climate data. Epidemics. [Internet]. 2025; 53:100854. doi: https://doi.org/qts5 DOI: https://doi.org/10.1016/j.epidem.2025.100854
Alasmari SMN, Tu CW, Khan M, Javed B, Liaqat I, Bahadar S, Altwaim SA, Chen CC, Junior ISV, Ali A. Impact of climate change on the tick-host-pathogen complex: distribution patterns, disease incidence, and host infestation. Rev. Bras. Parasitol. Vet. [Internet]. 2025; 34(4):e004725. doi: https://doi.org/qts7 DOI: https://doi.org/10.1590/s1984-29612025062
Ali A, Zahid H, Zeb I, Tufail M, Khan S, Haroon M, Tufail M, Bilal M, Hussain M, Alouffi A, Muñoz-Leal S, Labruna MB. Risk factors associated with tick infestations on equids in Khyber Pakhtunkhwa, Pakistan, with notes on Rickettsia massiliae detection. Parasit. Vectors. [Internet]. 2021; 14(1):363. doi: https://doi.org/qts8 DOI: https://doi.org/10.1186/s13071-021-04836-w
Ahmad I, Ullah S, Alouffi A, Almutairi MM, Numan M, Tanaka T, Chang SC, Chen CC, Ali A. First molecular- based confirmation of Dermacentor marginatus and associated Rickettsia raoultii and Anaplasma marginale in the Hindu Kush Mountain Range. Animals. [Internet]. 2023; 13(23):3686. doi: https://doi.org/qts9 DOI: https://doi.org/10.3390/ani13233686
Zhang T, Yan M, Fang J, Li X, Wang L, Wang H. Predicting future tropical theileriosis risk in China using tick distribution and climate models. Prev. Vet. Med. [Internet]. 2025; 244:106652. doi: https://doi.org/qttb DOI: https://doi.org/10.1016/j.prevetmed.2025.106652
Wang B, Liu Z, Zhu S, Zhang J, Qi W, Wang J, Li D, He L, Zhao J. Molecular characterization of bacterial and protozoan pathogens transmitted by ticks infesting livestock in Xinjiang, China. Parasit. Vectors. [Internet]. 2025; 18:207. doi: https://doi.org/qttc DOI: https://doi.org/10.1186/s13071-025-06857-1
Lansdell S, Sharif MS, Zorto A, Seto M, Negera E, Cutler S. Machine learning-based techniques for assessing critical factors for European tick abundance. Int. J. Comput. Theory Eng. [Internet]. 2025; 17(1):13-20. doi: https://doi.org/g9ccxg DOI: https://doi.org/10.7763/IJCTE.2025.V17.1364
Edwards RTM, Antoshechkin I, Hill E, Perry MW, Olafson PU, Saelao P, Lohmeyer KH, Akbari OS. First transcriptome analysis of the winter tick (Dermacentor albipictus) reveals sex-specific expression patterns and potential targets for genetic control. G3 (Bethesda). [Internet]. 2025; 15(8):jkaf116. doi: https://doi.org/qttd DOI: https://doi.org/10.1093/g3journal/jkaf116
Jouglin M, Bonsergent C, de la Cotte N, Mège M, Bizon C, Couroucé A, Lallemand ÉA, Leblond A, Lemonnier LC, Leroux A, Marano I, Muzard A, Quéré É, Toussaint M, Agoulon A, Malandrin L. Equine piroplasmosis in different geographical areas in France: prevalence heterogeneity of asymptomatic carriers and low genetic diversity of Theileria equi and Babesia caballi. Ticks Tick Borne Dis. [Internet]. 2025; 16(1):102434. doi: https://doi.org/g89pd7 DOI: https://doi.org/10.1016/j.ttbdis.2024.102434